- Your Product Type
- Your Study Type
- Aquatic Ecotoxicology
- Aquatic Invertebrates
- OECD 202: Daphnia sp., Acute Immobilisation Test
- OECD 211: Daphnia magna Reproduction Test
- OECD 235: Chironomus sp., Acute Immobilisation Test
- OECD 218/219: Sediment-Water Chironomid Toxicity Test Using Spiked Sediment/Spiked Water
- OECD 233: Sediment-Water Chironomid Life-Cycle Toxicity Test Using Spiked Water or Spiked Sediment
- OECD 225: Sediment-water Lumbriculus Toxicity Test Using Spiked Sediment
- OECD 242: Potamopyrgus antipodarum Reproduction Test
- OECD 243: Lymnaea stagnalis Reproduction Test
- Fish and other vertebrates
- OECD 203: Fish, Acute Toxicity Test
- OECD 215: Fish Juvenile Growth Study
- OECD 212: Fish, Short-term Toxicity Test on Embryo and Sac-fry Stages
- OECD 231: The Amphibian Metamorphosis Assay
- OECD 236: Fish Embryo Acute Toxicity Test
- OECD 210: Fish, Early-life Stage Toxicity Test
- OECD 229 Fish Short Term Reproduction Assay and OECD 230 21-day Fish Assay
- OECD 240 Medaka Extended One Generation Reproduction Test (MEOGRT)
- OECD 248: Xenopus Eleutheroembryonic Thyroid Assay
- OPPTS 850.1500: Fish Life Cycle Toxicity Test
- OÈCD 234 Fish sexual development test
- Aquatic plants
- Analytical Dose Verification
- Aquatic Invertebrates
- Chemistry
- Biodegradation Studies
- Analytical Chemistry Studies and Residues
- Physical-Chemical Properties Studies
- Storage Stability Studies
- OPPTS 830.6302, OPPTS 830.6303,and OPPTS 830.6304: Physical State, Colour and Odor at 20 °C and at 101.3 kPa
- EU A.1: Melting temperature/range
- EU A.2: Boiling temperature
- EU A.3: Relative density (liquids and solids)
- EU A.4: Vapour pressure
- EU A.5: Surface tension
- EU A.9: Flashpoint
- EU A.10: Flammability (solids)
- EU A.12: Flammability (contact with water)
- EU A.13: Pyrophoric properties of solids and liquids
- EU A.16: Relative self-ignition temperature for solids
- EU A.17: Oxidising properties
- OECD 114: Viscosity of Liquids
- Environmental Fate
- Transformation in Soil
- Transformation in Water
- Transformation in Manure
- Adsorption on Soil and Sewage Sludge
- Bioaccumulation and Bioconcentration
- Terrestrial Ecotoxicology
- Non-target Arthropods
- Non-target arthropod testing with the parasitic wasp (Aphidius rhopalosiphi)
- Non-target arthropod testing with the lacewing (Chrysoperla carnea)
- Non-target arthropod testing with the ladybird beetle (Coccinella septempunctata)
- Non-target arthropod testing with the predatory bug (Orius laevigatus)
- Non-target arthropod testing with the predatory mite (Typhlodromus pyri)
- Non-target arthropod testing with the rove beetle (Aleochara bilineata)
- Non-target arthropod testing with the carabid beetle (Poecilus cupreus)
- Non-target arthropod testing with the wolf spider (Pardosa spec.)
- Soil Organisms
- Honey Bees and other Pollinators
- OECD 213/214: Honey bees, Acute Oral and Acute Contact Toxicity Test
- OECD 245: Honey Bee (Apis Mellifera L.), Chronic Oral Toxicity Test (10-Day Feeding)
- OECD 237: Honey Bee Larval Toxicity Test, Single Exposure
- OECD 239: Honey Bee Larval Toxicity Test
- EPPO 170: Honey Bee Field Study – do plant protection products effect honey bee colonies?
- Oomen et al. 1992: Honey Bee Brood Feeding Study
- OECD 75: Honey Bee Brood Test under Semi-field Conditions in Tunnels
- OECD 246/247 Acute Oral and Contact Toxicity to the Bumblebee, Bombus terrestris L.
- Solitary Bee Acute Contact Toxicity Study in the Laboratory (Osmia sp.) Solitary Bee Acute Oral Toxicity Study in the Laboratory (Osmia sp.) (protocols for ringtests with solitary bees recommended by the non-Apis working group)
- SANTE/11956/2016 rev.9 Residue trials for MRL setting in honey
- Non-target plants
- OECD 208: Terrestrial Plant Test - Seedling Emergence and Seedling Growth Test
- OECD 227: Terrestrial Plant Test - Vegetative Vigour Test
- OCSPP 850.4100: Seedling Emergence and Seedling Growth
- OCSPP 850.4150: Vegetative Vigor
- EPPO PP 1/207(2): Efficacy evaluation of plant protection products, Effects on succeeding crops
- Field Studies
- Non-target Arthropods
- Ecological Modelling
- Quality Assurance
- Testing of Potential Endocrine Disruptors
- Aquatic Ecotoxicology
- News
- Company
- Career
- Contact
OECD 75: Honey Bee Brood Test under Semi-field Conditions in Tunnels
 It is important that plant protection products should be authorized for use only in ways which do not pose an unacceptable risk of harm to honey bees. In this context honey bee tests are an essential part of the environmental risk assessment except where plant protection products (ppp) are for exclusive use in situations where bees are not to be exposed. These honey bee brood tests are higher-tier studies and address concern regarding the impact on the brood development in honey bee colonies exposed to treatments of plant protection products.
It is important that plant protection products should be authorized for use only in ways which do not pose an unacceptable risk of harm to honey bees. In this context honey bee tests are an essential part of the environmental risk assessment except where plant protection products (ppp) are for exclusive use in situations where bees are not to be exposed. These honey bee brood tests are higher-tier studies and address concern regarding the impact on the brood development in honey bee colonies exposed to treatments of plant protection products.
They are also a reasonable approach, if a product is applied to the flowering crop and the active is known to be transferred into the bee hive in considerable amounts or a product is applied as a seed treatment or soil granule and the active is known to be a systemic compound found in pollen or nectar.
Study Design
Test organisms
Healthy honey bee colonies of appropriate size should be produced at the same time so that the colonies are as uniform as possible. The quantity of food stores within the hive at the beginning of a study should be kept at a minimum to ensure larvae being fed by contaminated food. This results in a maximum exposure of ppp`s to the bee brood.
Test Concentrations
Only formulated products are tested and should normally be applied at the highest field rate intended for the registration of the product in order to produce a worst-case exposure of the bees.
Course of the test
Each test should include 3 treatment groups with at least three replicates:
- Treatment with the plant protection product
- Treatment with a reference item known to produce adverse effects on honey bee brood (e.g. fenoxycarb)
- Control (treatment with tap water)
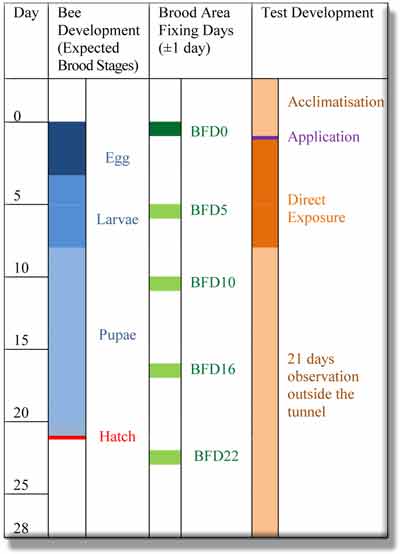
 The honey bee colonies are initially placed in tunnels (ca. 80 m2 crop size) containing an attractive flowering crop (e.g. Phacelia tanacetifolia). The bees are acclimatised in the tunnels for at least 3 days. Before the application day one brood comb with an appropriate amount of eggs is taken out of each colony and a digital picture of the comb is taken for the bee brood assessment (=BFD0; Brood Area Fixing Day). An area with at least 200 cells containing eggs are selected and marked by using a computer assisted digital program.
The honey bee colonies are initially placed in tunnels (ca. 80 m2 crop size) containing an attractive flowering crop (e.g. Phacelia tanacetifolia). The bees are acclimatised in the tunnels for at least 3 days. Before the application day one brood comb with an appropriate amount of eggs is taken out of each colony and a digital picture of the comb is taken for the bee brood assessment (=BFD0; Brood Area Fixing Day). An area with at least 200 cells containing eggs are selected and marked by using a computer assisted digital program.
Following BFD0 the test and reference item as well as the water control are applied to the flowering crop via spray application during bees actively foraging on the crop. For each subsequent brood assessment (BFDn) again the previously observed comb is taken out of the hive and another digital photo is taken in order to follow up the progress of the initial marked cells. This procedure is repeated in regular intervals (BFDn = 5, 10, 16, 22 days ± 1 day) until a full development of a bee is completed (e.g. 21 days). This allows a continuous photo-documentation following the first brood fixing day until the end of the assessment period (see table 1).
After 7 days of direct exposure of the colonies to the treated crop, the bee colonies are removed from the tunnels to an area with no main attractive crops. The full testing period is 28 days following BFD0.
Table 1: Bee Brood Assessments and Brood Stages (OECD 75)
Honey Bee Brood Assessment by Photo Documentation
 The honey bee brood assessment is conducted by photo documentation in combination with a computer assisted digital image analysis to improve the classical methods (acetate sheet method):
The honey bee brood assessment is conducted by photo documentation in combination with a computer assisted digital image analysis to improve the classical methods (acetate sheet method):
- Highest traceability and reliability for data verification
- A higher number of cells can be achieved which increases the reliability of the test
- Minimisation of impact on the bee brood by reducing the out of hive time (ca. 2-5 min)
- Automation of cell tracing excludes manual data transcription errors and improves reliability
- Possibility of re-analysation of images
For this purpose on each observation day the observed brood comb of a hive is reversibly positioned in a special constructed light box and a digital high resolution image is taken of the comb with a digital camera. Using an image analysing software for bee brood analysis (HoneyBee Brood Logger Software, supplier: WSC Scientific GmbH) it is possible to choose a high number of brood cells and to assess their development on the brood stage observation days given by table 1. Figure 1 shows an example of an image gallery of cells and their respective brood observation days +5, +11, +15 and +22. Their assessed brood stages are egg (BFD0), old larva (BFD5), pupa (capped cell) (BFD11 and BFD15) and empty cell or containig pollen (cell 7) (BFD22). All cells succesfully developed and hatched after 22 days.
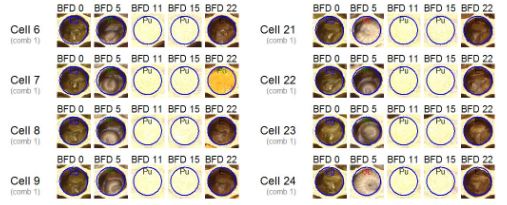
Figure 1: Photo documentation of cells (BFD0, BFD5, BFD11, BFD15 and BFD22)
Optional: Residue analysis
According to regulatory requirements residue studies may be required to determine residue levels in:
- pollen
- nectar
- flowers
- and/or foraging bees
In order to proove exposure, residue samplings can be incorporated into the semi-field study with additional hives used exclusevily for residue purposes. Bee hives used for residue samplings are not biologically assessed (e.g. mortality, foraging activity, colony assessments, etc.). Details about the residue samplings are described in the recent study protocol (e.g. timing, amount, etc.).
Endpoints / Parameters
- Meteorological data
- Foraging activity: in marked areas of 1 m² the number of bees are counted during 10-15 s.
- Mortality: sheets are spread out at the front, middle and back of the tunnels in order to facilitate the collection and counting of dead bees in the crop area. Additionally, mortality in the hives is assessed by counting dead bees in dead bee traps
- Behavioural abnormalities
- Condition of the colonies: the state of the adult bee population, the viability of the queen and the area of brood and food is assessed.
- Assessment of bee brood development
Guidelines and Literature
- OECD 75: “Guidance Document on the Honeybee (Apis Mellifera L.) Brood Test under Semi-Field Conditions"
- OEPP/EPPO guideline No. 170 (4) (OEPP/EPPO, 2010)
- AG Bienenschutz (2011)