- Your Product Type
- Your Study Type
- Aquatic Ecotoxicology
- Aquatic Invertebrates
- OECD 202: Daphnia sp., Acute Immobilisation Test
- OECD 211: Daphnia magna Reproduction Test
- OECD 235: Chironomus sp., Acute Immobilisation Test
- OECD 218/219: Sediment-Water Chironomid Toxicity Test Using Spiked Sediment/Spiked Water
- OECD 233: Sediment-Water Chironomid Life-Cycle Toxicity Test Using Spiked Water or Spiked Sediment
- OECD 225: Sediment-water Lumbriculus Toxicity Test Using Spiked Sediment
- OECD 242: Potamopyrgus antipodarum Reproduction Test
- OECD 243: Lymnaea stagnalis Reproduction Test
- Fish and other vertebrates
- OECD 203: Fish, Acute Toxicity Test
- OECD 215: Fish Juvenile Growth Study
- OECD 212: Fish, Short-term Toxicity Test on Embryo and Sac-fry Stages
- OECD 231: The Amphibian Metamorphosis Assay
- OECD 236: Fish Embryo Acute Toxicity Test
- OECD 210: Fish, Early-life Stage Toxicity Test
- OECD 229 Fish Short Term Reproduction Assay and OECD 230 21-day Fish Assay
- OECD 240 Medaka Extended One Generation Reproduction Test (MEOGRT)
- OECD 248: Xenopus Eleutheroembryonic Thyroid Assay
- OPPTS 850.1500: Fish Life Cycle Toxicity Test
- OÈCD 234 Fish sexual development test
- Aquatic plants
- Analytical Dose Verification
- Aquatic Invertebrates
- Chemistry
- Biodegradation Studies
- Analytical Chemistry Studies and Residues
- Physical-Chemical Properties Studies
- Storage Stability Studies
- OPPTS 830.6302, OPPTS 830.6303,and OPPTS 830.6304: Physical State, Colour and Odor at 20 °C and at 101.3 kPa
- EU A.1: Melting temperature/range
- EU A.2: Boiling temperature
- EU A.3: Relative density (liquids and solids)
- EU A.4: Vapour pressure
- EU A.5: Surface tension
- EU A.9: Flashpoint
- EU A.10: Flammability (solids)
- EU A.12: Flammability (contact with water)
- EU A.13: Pyrophoric properties of solids and liquids
- EU A.16: Relative self-ignition temperature for solids
- EU A.17: Oxidising properties
- OECD 114: Viscosity of Liquids
- Environmental Fate
- Transformation in Soil
- Transformation in Water
- Transformation in Manure
- Adsorption on Soil and Sewage Sludge
- Bioaccumulation and Bioconcentration
- Terrestrial Ecotoxicology
- Non-target Arthropods
- Non-target arthropod testing with the parasitic wasp (Aphidius rhopalosiphi)
- Non-target arthropod testing with the lacewing (Chrysoperla carnea)
- Non-target arthropod testing with the ladybird beetle (Coccinella septempunctata)
- Non-target arthropod testing with the predatory bug (Orius laevigatus)
- Non-target arthropod testing with the predatory mite (Typhlodromus pyri)
- Non-target arthropod testing with the rove beetle (Aleochara bilineata)
- Non-target arthropod testing with the carabid beetle (Poecilus cupreus)
- Non-target arthropod testing with the wolf spider (Pardosa spec.)
- Soil Organisms
- Honey Bees and other Pollinators
- OECD 213/214: Honey bees, Acute Oral and Acute Contact Toxicity Test
- OECD 245: Honey Bee (Apis Mellifera L.), Chronic Oral Toxicity Test (10-Day Feeding)
- OECD 237: Honey Bee Larval Toxicity Test, Single Exposure
- OECD 239: Honey Bee Larval Toxicity Test
- EPPO 170: Honey Bee Field Study – do plant protection products effect honey bee colonies?
- Oomen et al. 1992: Honey Bee Brood Feeding Study
- OECD 75: Honey Bee Brood Test under Semi-field Conditions in Tunnels
- OECD 246/247 Acute Oral and Contact Toxicity to the Bumblebee, Bombus terrestris L.
- Solitary Bee Acute Contact Toxicity Study in the Laboratory (Osmia sp.) Solitary Bee Acute Oral Toxicity Study in the Laboratory (Osmia sp.) (protocols for ringtests with solitary bees recommended by the non-Apis working group)
- SANTE/11956/2016 rev.9 Residue trials for MRL setting in honey
- Non-target plants
- OECD 208: Terrestrial Plant Test - Seedling Emergence and Seedling Growth Test
- OECD 227: Terrestrial Plant Test - Vegetative Vigour Test
- OCSPP 850.4100: Seedling Emergence and Seedling Growth
- OCSPP 850.4150: Vegetative Vigor
- EPPO PP 1/207(2): Efficacy evaluation of plant protection products, Effects on succeeding crops
- Field Studies
- Non-target Arthropods
- Ecological Modelling
- Quality Assurance
- Testing of Potential Endocrine Disruptors
- Aquatic Ecotoxicology
- News
- Company
- Career
- Contact
OECD 208: Terrestrial Plant Test - Seedling Emergence and Seedling Growth Test
Non-target plants, i.e. plants in natural and semi-natural habitats within the agricultural land such as hedgerows, field borders, meadows and other small biotopes may unintentionally be exposed to Plant Protection Products, veterinary medicinal products, biozides or chemicals.
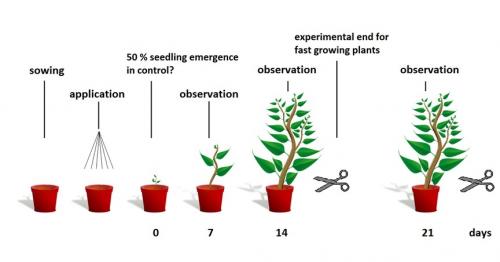
The OECD guideline 208 assesses the potential effects of a test item on seedling emergence and early growth of seedlings following exposure to the test item in or on the soil.
Study Design
Plant species
The plant species selected for the test should derive from different plant families to consider the taxonomic diversity and should give reliable and reproducible results. Usually 6 to 10 plant species are chosen according to the specific properties of the test item and the intended use. Among them could be for example Allium cepa (onion), Zea mays (corn), Brassica napus (oilseed rape), Cucumis sativus (cucumber), Glycine max (soybean), Helianthus annuus (sunflower), Solanum lycopersicon (tomato) or Stellaria media (common chickweed).
Test concentrations / rates
Test concentrations / rates are usually determined in a range-finding pre-test. For plant protection products the rates for the pre-test are calculated based on the maximum application rate. For veterinary medicinal products concentrations for the pre-test can be calculated based on the expected PEC (Predicted Environmental Concentration) soil. Alternatively, it is possible to use 1000 mg/kg dry soil as maximum concentration in the pre-test.
Course of the test
Depending on the expected route of exposure, the test item is either sprayed on to the soil surface after sowing (plant ptrotection products) or the test item is incorporated into the soil (veterinary medicinal products, chemicals) and seeds are planted in this treated soil. The test item concentration has to be confirmed analytically. After the application the plants are evaluated weekly for effects (emergence and phytotoxticity) in comparison to the water treated control plants for 14 to 21 days after 50 % emergence of the seedlings in the control group. At the test end the endpoints are measured and recorded.
Seedling emergence and seedling growth test with test item treated manure for veterinary medicinal products
In the case of strong effects for a veterinary medicinal products in a OECD 208 test this test item can be applied to manure to have a more realistic approach (Concept development for an extended plant test in the environmental risk assessment of veterinary medicinal products). This manure is then incubated under anaerobic conditions. The incubation time depends on the type of manure (cattle, pig) and its normal storage time. The spiked manure is then incorporated into the soil. The further study design has to fullfill the requirements of OECD 208. The effects are evaluated in comparison to a non-spiked manure treated control.
Growth Conditions
The studies are performed in a growth chamber with controlled, optimized test conditions for standardised studies. The temperature is 22 °C during the light period and 18 °C during the dark period (mean 22 °C ± 10 °C) and humidity is 70% ± 25%. During the photoperiod of 16 hours light the intensity is 350 ± 50 µE/m2/s.
For special needs, more differentiated studies and research these conditions can be adapted within a wide range.
Endpoints
Endpoints are emergence, mortality and shoot fresh weight. If the test is conducted as multiple rate test, effects on fresh weight are reported as ERX (Effective Rate) for each species individually as NOER (No Observed Effect Rate) in case of a limit test.
Additional parameters are shoot height and shoot dry weight, reported as ERX for each species individually.
Phytotoxicity (e.g. chlorosis, necrosis, deforamtion) and growth stages (BBCH code) are recorded. If the test is conducted as multiple rate test an ERX (effective rate) caluation on phytotoxicity is possible.
Guidelines and literature
-
OECD Guidelines for Testing of Chemicals No. 208 “Terrestrial Plant Test: Seedling Emergence and Seedling Growth Test”.
-
Concept development for an extended plant test in the environmental risk assessment of veterinary medicinal products, Federal Enviroment Agency.